
One key property of metalloids is their ability to conduct electricity under certain conditions.įor example, silicon is a semiconductor, meaning it can be used to control the flow of electricity in electronic devices. Īrsenic has been used in various applications including wood preservatives and insecticides, although it is now recognized as a toxic substance. 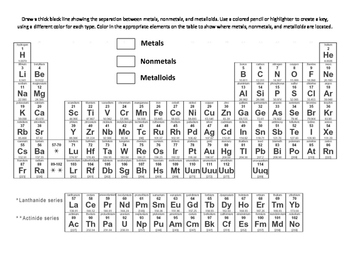
Metalloids tend to have properties that make them useful in technological and industrial applications.įor example, silicon is an important component of semiconductors, while boron is used in the production of strong and lightweight alloys. They are located along the “staircase” that separates the two categories on the periodic table. Metalloids are elements that share properties with both metals and nonmetals. Nonmetals also tend to form negative ions when they react with metals, while metals tend to form positive ions. Nonmetals have several physical and chemical properties that distinguish them from metals.įor example, nonmetals tend to be poor conductors of heat and electricity, and they have lower melting and boiling points than metals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
/periodic-table--illustration-738787291-59888b4822fa3a00109a466d.jpg)
 RSS Feed
RSS Feed
